Ancient Zoonotic Pathogens in Animal Bones: Integrating Zooarchaeology, Palaeopathology and DNA Across 6,000 Years




Ancient Animal Pathogens: Hunting Disease in Archaeological Bones
This comprehensive study examines animal bone collections from 34 archaeological sites across Eurasia, spanning roughly 6,000 years of human-animal cohabitation. The research addresses a fundamental question in archaeological science: where exactly should researchers drill to find the DNA of ancient diseases? Rather than randomly sampling bones, this investigation employs a targeted strategy, guided by visible traces of illness on skeletal remains and carefully selected teeth from domestic and wild animals.
The methodology represents a significant advancement in zooarchaeological pathogen detection, demonstrating how visible skeletal disease can serve as a powerful guide for prioritizing which specimens warrant the considerable cost and effort of ancient DNA analysis. By focusing on bones that clearly show signs of past illness, researchers dramatically increase their chances of recovering authentic ancient pathogen DNA while advancing our understanding of disease ecology in prehistoric communities.
The research team assembled 346 skeletal elements from at least 328 individual animals, drawn from sites spanning approximately six thousand years of Eurasian prehistory and history. The temporal range extends from the Neolithic settlement of Monjukli Depe in Turkmenistan, dating to approximately 4650-4350 BCE, through to the early medieval site of Giecz 10 in Poland, occupied between 900-1200 CE. This extensive chronological framework allows researchers to trace the evolution of animal-borne diseases across multiple cultural and technological transitions.
The sampling strategy deliberately favored animals that held central importance in ancient economies: cattle, sheep, pigs, goats, and dogs. Sheep dominate the pathological collection with 68 of the 187 bones showing clear signs of disease, but the assemblage also includes horses, red deer, aurochs, gazelles, a fox, and even a beaver. These animals represent the working beasts, food sources, and companions of Bronze Age and later communities, serving as crucial potential reservoirs of infection that could affect human populations.
Many specimens derive from Bronze Age contexts, that pivotal period of large-scale migrations and evolving lifeways when humans and their domestic animals were living in unprecedented proximity. Sites from this era include Tilla Bulak in Uzbekistan, Marinskaya 5 in the North Caucasus, Pietrele in Romania, and various locations around Augsburg in Germany. These settlements represent diverse ecological and cultural contexts where intensive animal husbandry created new opportunities for pathogen transmission between species.
The research team employed sophisticated paleapathological analysis, carefully examining animal remains from several key regions including Germany, Poland, Romania, and Uzbekistan. They sought telltale signs of ill health preserved in the skeletal record, focusing on changes that could reflect chronic infection or inflammatory disease processes. The selection process yielded 188 bones showing potential evidence of disease, 27 apparently healthy bones with no visible lesions, and 131 teeth for comparative analysis.
The pathological bones originated from various archaeological contexts including refuse pits, settlement layers, and burial deposits. These specimens were often preserved in sand or silt matrices, sometimes encrusted with mineral deposits like gypsum. Researchers carefully assessed taphonomic changes such as weathering, carnivore gnawing, and trampling to distinguish genuine disease markers from post-depositional damage.
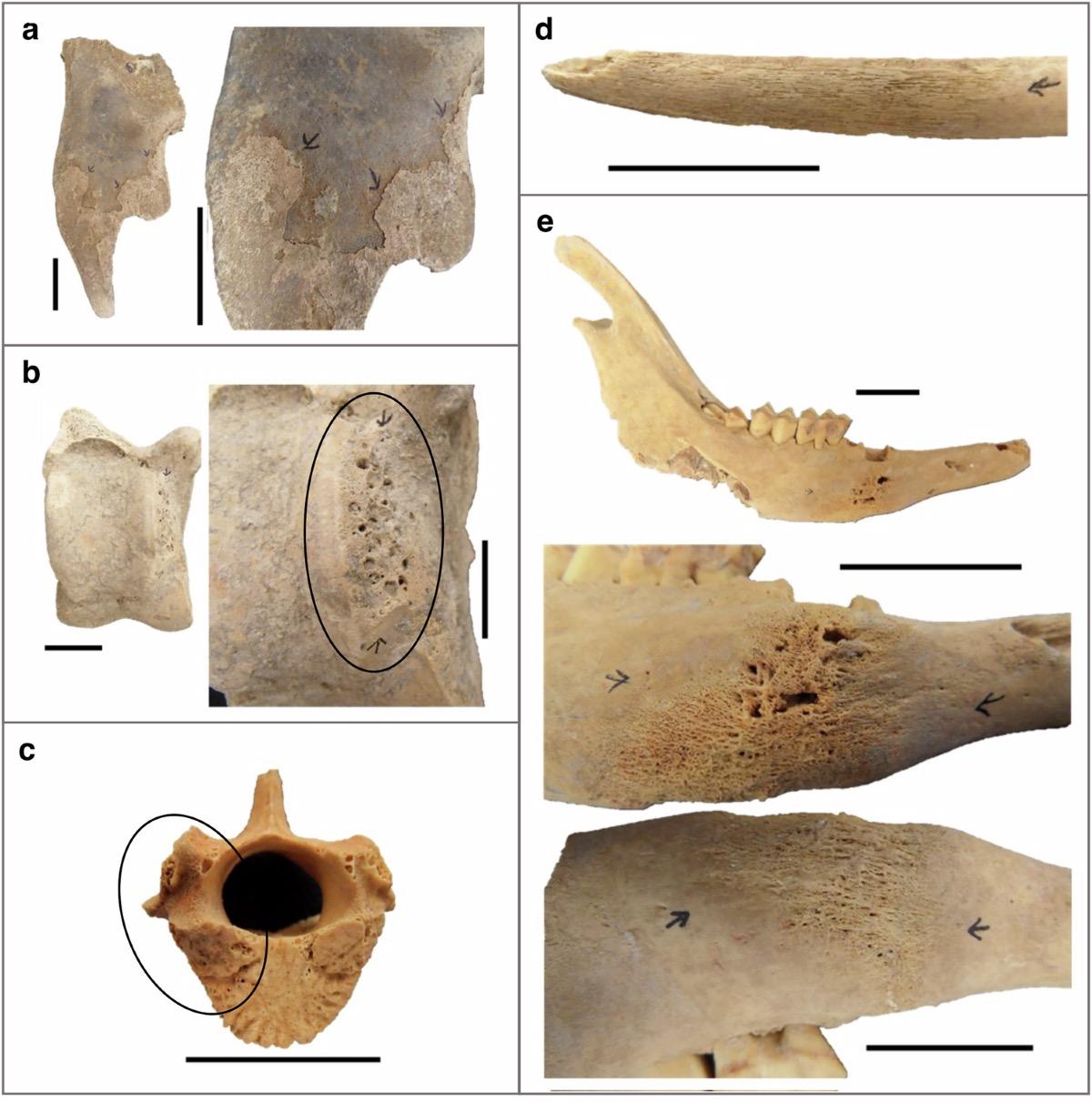
Specific pathological indicators included porous and roughened bone surfaces suggesting chronic inflammation, swollen and misshapen joints indicating arthritis or septic arthritis, reactive new bone formation around teeth signaling oral infections, and areas of bone destruction potentially caused by abscesses or other infectious processes. Because most archaeological animal bones are fragmentary and disarticulated, researchers typically examine individual elements rather than complete skeletons, making diagnosis challenging since many lesions are non-specific and heavy labor can produce changes that mimic disease processes.
Alongside diseased bones, researchers sampled 131 teeth from most sites in the study. Teeth serve as particularly valuable repositories for ancient DNA because they can protect genetic material within their inner pulp chambers. In human studies, teeth have proven to be reliable sources of blood-borne pathogen genomes, making them attractive targets for ancient disease research.
However, animal dental anatomy complicates this picture significantly. Dogs and pigs, apart from their tusks, possess short-crowned teeth with enclosed pulp chambers similar to human dentition. In contrast, cattle, sheep, and goats have high-crowned teeth with open roots and less clearly contained pulp spaces. The research team was uncertain whether these different dental architectures would preserve pathogen DNA equally well, so they deliberately included both types across multiple species and sites to test preservation patterns.
The sampling methodology was refreshingly straightforward. Paleapathological lesions including rough, swollen, porous, or pitted patches suggesting past illness were drilled specifically, often sampling across the surface of lesions and then deeper into underlying spongy bone tissue. When possible, trabecular bone away from visible lesions was also collected to detect any systemic bloodstream infections that might have seeded the skeleton more broadly.
Teeth were treated as individual micro-sites for sampling purposes. Researchers sawed off tooth roots and drilled into both the root body and, where accessible, the interior pulp space. Small or fragile deciduous teeth, particularly from dogs, were powdered completely for analysis. For several German mandibles, the team could directly compare teeth and jawbone from the same individual animal, allowing precise comparison of different skeletal elements as pathogen DNA sources.
The 346 skeletal elements ultimately yielded 357 separate DNA extracts, with some bones sampled at multiple locations and several individuals contributing both bones and teeth to the study. DNA extraction and sequencing followed strict contamination protocols in dedicated ancient DNA laboratories, with careful authentication procedures to distinguish genuine ancient pathogen DNA from modern contamination.
Once sequenced and screened for bacterial DNA signatures, the research revealed clear benefits of the selective sampling strategy. Of all DNA libraries produced, 55 yielded robust signals of bacterial pathogens displaying the characteristic damage patterns expected of authentic ancient DNA. These positive results included 44 samples from bones with visible pathological lesions, 11 from teeth, and notably, zero from apparently healthy bones without visible disease markers.
One of the most significant discoveries involved Erysipelothrix rhusiopathiae, a bacterium that today causes swine erysipelas in pigs and severe infections in cattle, and can also infect humans handling animals or meat products. The research team identified this pathogen in two Bronze Age specimens: a cattle tooth from Marinskaya 5 in the North Caucasus and a cattle astragalus (ankle bone) from Pietrele showing coarse, porous bone suggestive of chronic inflammation.
Phylogenetic analysis revealed that this Bronze Age bovine infection represents an early branch of the species, positioned basally to modern diversity and clustering with medieval human cases from Iberia. This finding suggests a long-standing, multi-host evolutionary history for the pathogen, demonstrating that it was already circulating widely in Bronze Age livestock across Eurasia and potentially infecting humans who worked closely with these animals.
The study identified Streptococcus lutetiensis, a bacterium currently recognized as a cause of mastitis in dairy cattle, in several Bronze Age animals from Tilla Bulak. Despite relatively low genomic coverage, these ancient strains form a distinct clade that branches off before known modern strains, suggesting a deep Bronze Age ancestry for infections associated with milk-producing herd animals.
Three specific animals from Tilla Bulak carried this pathogen: two sheep and one goat, all showing skeletal evidence of chronic stress and disease. The presence of mastitis-causing bacteria in Bronze Age flocks indicates that udder infections were already affecting milk production and animal welfare over 3,500 years ago, representing a significant economic and health challenge for ancient pastoral communities.
Several individual animals provided vivid examples of chronic disease processes. A sheep mandible from Pietrele displayed dramatic pathological changes including heavy reactive bone formation, abscess development, and early fistula formation. DNA analysis revealed multiple bacteria associated with oral infection and inflammation, including Streptococcus lutetiensis and various Staphylococcus species, demonstrating the complex polymicrobial nature of ancient infections.
Dog specimens yielded evidence of zoonotic pathogens including Bordetella petrii, related to modern respiratory pathogens affecting both animals and humans, and Coxiella burnetii, the causative agent of Q fever. These findings demonstrate that companion animals were fully integrated into Bronze Age disease networks, sharing pathogens with both livestock and human populations in close-knit household environments.
The Bronze Age settlement of Tilla Bulak in present-day Uzbekistan emerged as an exceptional source of ancient pathogen DNA. Contributing 105 specimens representing approximately 29% of the total dataset, this site produced more than half of all positive pathogen detections in the study. Many specimens were sheep and goat bones displaying obvious inflammatory changes, with several yielding sufficient pathogen DNA for placement within evolutionary frameworks alongside modern bacterial strains.
The concentration of positive results at Tilla Bulak likely reflects both favorable preservation conditions and the intensive animal husbandry practices of this Bronze Age oasis community. The site's location along ancient trade routes and its role as a center for pastoral activities may have created ideal conditions for pathogen circulation among dense animal populations living in close proximity to human settlements.
The Polish Bronze Age site of Inowrocław demonstrated relatively good preservation of host DNA and achieved higher pathogen detection rates than expected from its modest sample size. In contrast, the older Chalcolithic site of Pietrele on the Lower Danube, though rich in pathological bones and contributing 23% of total specimens, produced fewer pathogen identifications, consistent with its greater antiquity and more challenging preservation environment.
These patterns suggest that both ancient treatment of animal remains including butchery, cooking, and disposal practices, and depositional environment factors such as sediment chemistry, moisture levels, and soil conditions, significantly influenced pathogen DNA survival. However, within any given site, bones displaying visible disease markers consistently proved most promising for ancient pathogen recovery.
The research demonstrates that visible skeletal disease provides an effective screening method for identifying specimens most likely to contain preserved pathogen DNA. Statistical analysis confirmed that bones with lesions significantly outperformed apparently healthy specimens in pathogen detection rates, validating palaeopathology as a practical tool for ancient DNA research prioritization.
Authentication of ancient pathogen DNA relied on multiple criteria including characteristic ancient DNA damage patterns, appropriate phylogenetic placement relative to modern strains, and consistency with archaeological and palaeopathological evidence. This multi-layered approach helps distinguish genuine ancient infections from modern laboratory contamination, a persistent challenge in ancient DNA research.
The study's success rate varied considerably among specimen types, with mandibles, ribs, and vertebrae proving particularly productive sources of pathogen DNA. These skeletal elements are commonly preserved in archaeological contexts and frequently display pathological changes associated with oral infections, respiratory disease, and spinal inflammation, making them ideal targets for future ancient pathogen research.
The findings provide direct evidence for the antiquity of several important zoonotic pathogens, demonstrating that many bacteria now associated with livestock disease and human infection were already established in Bronze Age animal populations. Phylogenetic analyses place ancient strains at basal positions within modern bacterial diversity, suggesting that current pathogen diversity represents recent radiation from much older lineages.
The presence of Salmonella enterica in both Bronze Age and earlier contexts supports previous hypotheses linking the emergence of human-adapted Salmonella lineages to the spread of agriculture and intensive animal husbandry in Eurasia. Similarly, evidence for Brucella and Yersinia species in archaeological contexts demonstrates the deep history of these significant human pathogens.
This research contributes to an emerging "One Health" approach to archaeological science, recognizing that human health has always been intimately connected to animal health and environmental conditions. The study demonstrates how ancient settlements functioned as integrated disease landscapes where pathogens circulated freely among humans, domestic animals, and wild species.
Bronze Age communities like those at Tilla Bulak and Pietrele created unprecedented opportunities for pathogen transmission through intensive animal husbandry, shared living spaces, and complex networks of trade and migration. The resulting disease ecology established many of the zoonotic pathogen lineages that continue to affect human and animal health in modern agricultural and urban environments.
The targeted sampling strategy developed in this research offers a practical framework for expanding ancient pathogen studies to additional archaeological contexts and time periods. By focusing on bones with visible pathological changes, researchers can maximize the efficiency of expensive ancient DNA analyses while building larger datasets for understanding disease evolution and emergence.
The study also demonstrates the value of interdisciplinary collaboration between archaeologists, palaeopathologists, and geneticists in reconstructing ancient disease landscapes. This integration of evidence from skeletal analysis, archaeological context, and molecular biology provides unprecedented resolution in understanding how pathogens shaped ancient human-animal relationships.
Ultimately, this research transforms our understanding of archaeological animal remains from simple economic indicators to complex archives of ancient disease ecology. The bones and teeth recovered from refuse pits and settlement layers preserve not just evidence of diet and subsistence practices, but detailed records of the pathogens that circulated among ancient communities and continue to influence modern disease patterns.
This comprehensive investigation demonstrates that ancient animal bones, when carefully selected and analyzed, serve as remarkable archives of prehistoric disease ecology. The targeted sampling strategy based on visible pathological lesions significantly improves pathogen DNA recovery rates while providing insights into the deep evolutionary history of important zoonotic bacteria.
The research reveals that many pathogens now associated with modern livestock disease and human infection were already established in Bronze Age animal populations, circulating in the same intensive agricultural systems that characterize much of contemporary food production. These findings underscore the importance of understanding disease history for addressing current and future challenges in global health, food security, and pandemic preparedness.
By treating archaeological animal remains as direct archives of ancient infections rather than merely economic debris, this study opens new avenues for understanding how human-animal-pathogen relationships have evolved over millennia. The bones recovered from ancient settlements preserve not just evidence of past lifeways, but detailed molecular records of the diseases that shaped the health and survival of both human and animal communities throughout the development of agricultural civilization.
Original source: https://www.nature.com/articles/s41467-026-71543-4
Comments